Description
Red Cabbage (Brassica Oleracea L.) Extract Powder
Whether or not you like to eat red cabbage, you are going to love experimenting with it. This dried cabbage powder will allow you to make your own red cabbage pH indicator. This is a very magical kitchen science activity.
Making a red cabbage indicator is a fantastic STEAM activity to introduce children to acid/base chemistry.
- 10 g dried cabbage powder
- pH colour card
Red cabbage – which is purple! – is a natural acid/base indicator
- Contents makes1 L of red cabbage pH indicator.
- Want colours darker? Use less water.
- Want colours lighter? Use more water
Instructions
- Add half of the sachet contents to 500 ml of warm water. Let the juice cool.
- Your juice is now ready to experiment with!
- If you want the colours to be darker add more powder; add more water if you want the colours to be lighter.
If you don’t use it all – freeze it. Scientific Sue pours hers into ice cube trays and then uses one cube at a time as and when she needs them.
What is red cabbage?
Red cabbage is a cruciferous vegetable of firmly packed dark red-purple leaves. It belongs to the brassica group of vegetables along with Brussels sprouts and kale, and has a peppery taste and crunch when eaten raw, and becomes sweeter and softer in texture when cooked.

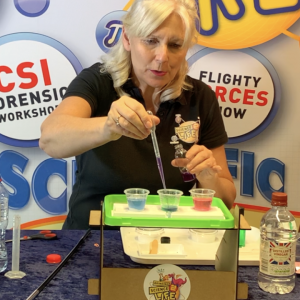
Red cabbage pH indicator experiment showing colour changes from acid to alkali using common household items.
Red cabbage is grown in Europe and is in season from September to December. As the plants grow, they form tight balls of leaves in the centre surrounded by much larger green-purple leaves. When the red cabbage is ready for harvesting, the whole plant is picked and the outer leaves discarded, leaving just the cabbage head – the part we eat.
Red cabbage has a good mix of vitamins and minerals, especially folate, which is essential during pregnancy and also helps the body to produce red blood cells. It also contains vitamin C, which helps protect our cells by acting as an antioxidant, and potassium, which we need for a healthy heart.
A 2019 study indicates growing evidence that anthocyanins play a positive role in cardiovascular health and that those who eat foods with anthocyanins have a lower risk of heart attacks and heart disease-related death.
For Parents & Teachers
“Safe Science at Home: Understanding pH with Red Cabbage”
“Teaching pH Safely: A Colourful Journey Through Household Chemistry”
For those interested in the science behind the magic, I’ve created a 24-minute educational video that dives deeper into the chemical reactions, the household substances used in the red cabbage pH scale, and—most importantly—how to stay safe while experimenting. 🧪✨
🎥 Watch it here: https://www.youtube.com/watch?v=gQYd4H3-ZxY
Perfect for curious parents, teachers, or anyone guiding young scientists through hands-on learning!
Science in a Nutshell
The purple colour in the red cabbage comes from a class of pigments called anthocyanins; this pigment is also found in the skin of red apples, grapes, plums and is the pigment in leaves which turn red in the autumn.
When the vinegar (ethanoic acid) is mixed with the baking soda (sodium hydrogen carbonate – a BASE) a chemical reaction takes place and the end solution will be a purple colour – indicating neutralisation has taken place.
Note if the baking soda it dissolved in water it then becomes an ALKALI.
Beware LOTS of bubbles of carbon dioxide are formed; these bubbles are as a result of the reaction – the bubbles will more than likely cascade over the top of the beakers – carry this experiment out on a tray or over a bucket!
Acidity is measured on a pH scale which runs from 0 (most acidic) to 14 (most basic or alkaline).
A substance which is neither acidic nor basic is called ‘neutral’ and has a pH of 7.
An acid solution contains an excess of hydrogen ions (H+) i.e. pH is a measure of how acidic a solution is or how many H+ ions are present.
Alkaline solutions have a pH greater than 7 meaning they have less free H+ ions than that of neutral water.
Citric acid crystals and liquid vinegar are both called acids.
Acids react with a base (solid) and an alkali (liquid) to produce a salt, water and carbon dioxide gas.
Safety: Due to the high likely hood of splashes being made younger scientists should wear safety or swimming googles.
Citric acid, baking soda and soap are the key ingredients found in BATH BOMBS!