Description
Citric Acid (125 g)
Sour, safe (when used correctly), and full of science!
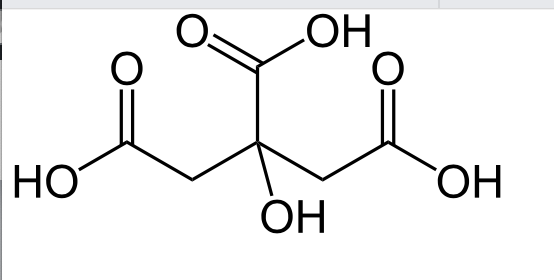
Citric acid is a naturally occurring weak organic acid found in citrus fruits such as lemons and limes. In the European Union it is approved as a food additive and is known as E330.
Although it looks like sugar, don’t be fooled — it definitely doesn’t taste sweet! Citric acid gives many favourite sour treats their sharp tang, including lemon drops, sour gummies, and “toxic waste” sweets.
But for Scientific Sue, citric acid isn’t just about flavour…
It’s all about the science! 🔬
Why is citric acid so useful in science activities?
Citric acid is one of the stronger edible acids, which makes it perfect for safe, exciting experiments at home or in the classroom.
It can be used to:
✨ Make fizzing bath bombs
✨ Create colour-changing bubble baths
✨ Produce carbon dioxide gas in exciting reactions
✨ Explore acids and alkalis
✨ Investigate chemical reactions and gas formation
✨ Make refreshing homemade lemonade
It even appears in Scientific Sue’s magical demonstrations — including the famous Amortentia Love Enchantment from her Magical Science shows! 🧪✨
The science behind the fizz
When citric acid reacts with bicarbonate of soda, the two substances combine to release carbon dioxide gas. This is what creates the bubbles, fizzing, and excitement in bath bombs and many classic science experiments.
A simple ingredient… with powerful chemistry!
The science behind the fizz
When citric acid reacts with bicarbonate of soda, the two substances combine to release carbon dioxide gas. This is what creates the bubbles, fizzing, and excitement in bath bombs and many classic science experiments.
A simple ingredient packed with powerful chemistry — helping you bring Science2Life through fizzing reactions, colour changes, and hands-on discovery.
Safety information
Citric acid is a weak acid, but like all science materials it should be handled sensibly.
Please note:
• Avoid breathing in the powder
• Avoid contact with eyes
• Wash hands after use
• Do not taste unless used as part of a supervised food activity
• Store in a dry place and keep out of reach of young children
Contact with concentrated powder or solutions may cause:
• eye irritation
• skin irritation
• coughing or sore throat if inhaled
• enamel erosion if consumed frequently in strong concentrations
Always use as directed during supervised science activities.